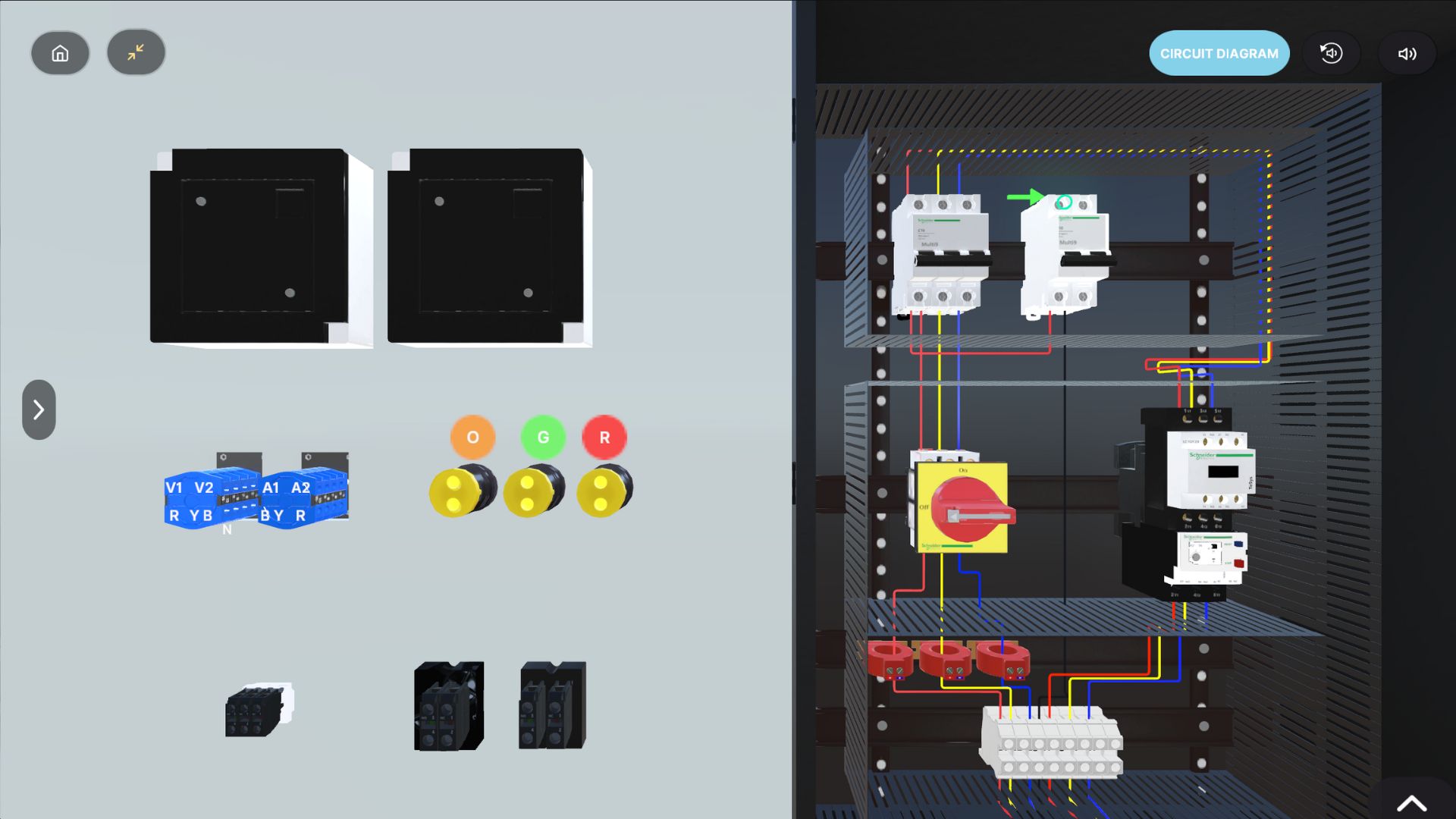
About Our Solutions
Pharmaceutical operations demand strict discipline. A small miss in procedure can lead to contamination risk, batch loss, audit findings, or safety incidents. Spatio builds VR training simulators that let teams practice critical GMP workflows and high-risk protocols in a safe, controlled environment, without disrupting production.
These modules support operators, supervisors, QA, and lab teams with repeatable practice that improves SOP adherence and confidence on the floor.
Real-World Scenarios
Train GMP workflows and cleanroom routines with realistic procedures
Compliance Assured
Reinforce SOP discipline, documentation accuracy, and audit readiness
Improved Retention
Repetition and assessments strengthen recall and reduce deviations
Global Scaling
Deploy across plants with centralized tracking and multilingual support
Custom VR Modules for the Pharma Industry
Comprehensive VR modules designed for pharmaceutical manufacturing and quality operations.
Pharmaceutical Process Training Modules

Line Changeover Training
Practice line clearance, tool segregation, equipment checks, status labeling, and restart verification to prevent cross contamination and deviations.

Feed Mix Preparation Training
Train weighing discipline, raw material verification, sequencing, mixing controls, sampling steps, and documentation for consistent batch outcomes.

HPLC and GCMS Drug Inspection Training
Practice sample preparation, method adherence, instrument handling basics, result review steps, and reporting accuracy for inspection readiness.

Aseptic Protocols Training
Rehearse gowning, hand hygiene, cleanroom movement, sterile transfers, contamination controls, and intervention discipline for sterile manufacturing areas.

API Safety Training for Harsh Chemicals
Train safe handling, PPE selection, exposure controls, spill response, waste segregation, and decontamination for high-risk API environments.

Deviation Response and CAPA Discipline
Train containment steps, documentation accuracy, escalation workflow, root cause thinking, and CAPA follow-up to strengthen audit outcomes.
Who We Are
Spatio builds custom VR simulators for compliance-driven, high-risk industries. We model your GMP workflows, cleanroom zones, SOP steps, and quality checkpoints so training matches real work. The goal is measurable: fewer deviations, stronger audit readiness, and safer handling practices.
Impact of our work
60%
Improvement in SOP adherence
43%
Reduction in safety incidents
75%
Increase in knowledge retention
70%
Reduction in training costs
Contact Us
Case Studies
What Our Clients Say
“Inspection routines became consistent across analysts. Supervisors coached gaps using clear assessments, and documentation quality improved.”
QA Manager
Pharmaceutical Quality Lab
Frequently Asked Questions
Yes. Spatio mirrors your zones, workflows, equipment steps, and documentation routines so training transfers directly to your plant.
Spatio is device agnostic. Modules run on VR headsets, and multi-user training can also be delivered via phone and desktop.
Yes. Multi-user modules allow trainees and trainers to connect in the same session for guided learning and coordinated practice.
Dashboards show completions, assessments, errors, and time to competency. Reports can export and integrate with your LMS.
No. Spatio pricing is not user-based licensing. Programs are designed for enterprise rollouts without per-user limits.
Ready to reduce deviations and strengthen audit readiness with VR training?
News & Insight
We share the latest breakthroughs, innovations, andupdates in the XR & AI industry